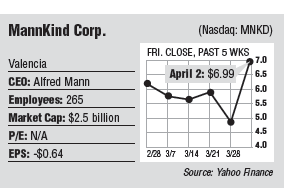
After more than a decade of constant losses and massive investments from its billionaire founder just to stay afloat, MannKind Corp. is one step away from hitting the jackpot. Investor confidence in the Valencia biotech has never been higher, with the stock price doubling in after-hours trading last week following an announcement that a Food and Drug Administration advisory panel in Washington, D.C. recommended approval of its first pipeline drug, Afrezza, an inhalable insulin system. Shares rose more than 100 percent after the April 1 announcement, but then lost some ground the next day. Overall, they were up 74 percent to $6.99 at the close of trading April 2. The gains helped medical device entrepreneur Alfred Mann, who has steered the company toward its goal for more than a decade, get his money back. It’s estimated that he has sunk about $900 million into the firm, and his 40 percent stake was worth about $1 billion as of April 2. Of course, the gains are only on paper, and the stock could move backwards. But if the company were to receive final FDA approval and reach an agreement with a larger pharmaceutical company to market the drug, analysts have pegged sales of the inhalable insulin at $1 billion to $4 billion annually. That would mean much bigger gains for the stock. “We’re in discussions with several large companies for marketing,” Chief Financial Officer Matthew J. Pfeffer told the Business Journal. “Approval is the next step, so we’re not able to speculate on a timeline when we would pick a partner. But clearly, sooner is better.” Attempts to contact Mann, 88, were unsuccessful, but in a statement last week he said, “Diabetes is a major health problem in the United States, and we are committed to bring Afrezza to the many patients who might benefit from this novel product.” Before commercialization is even a question, MannKind needs to get the final go-ahead from the FDA, which is expected in the next week. The agency isn’t required to follow the panel’s recommendation when it makes its decision, though that doesn’t happen often, said Graig Suvannavejh, senior health care analyst at New York investment bank MLV & Co. However, the FDA has rejected the drug twice before, most recently in 2011, asking the company for more data and clinical trials on its revamped inhaler, which was not part of the original application in 2009. “There’s a very small chance the FDA will go against the advisory panel,” said Suvannavejh. “It has happened a handful of times, but it’s a low figure.” Insulin replacement? If Afrezza can get the final OK, work its way through post-approval testing and reach the commercial sector, it would be the only inhaled insulin product on the market. In 2006, pharmaceutical giant Pfizer Inc. of New York received approval for its Exubera inhaler, but the company discontinued the product about a year later, citing weak sales. That led competitors Eli Lilly & Co. of Indianapolis and Novo Nordisk in Denmark to give up research on inhalable insulin, though one of those firms could easily reach an agreement with MannKind to help market Afrezza. “MannKind is not going to be able to do this on their own,” said Suvannavejh. “When you get in a situation where a drug needs to be prescribed, you need large commercial backing and marketing budgets. You need something on a global scale.” When the FDA approved Pfizer’s Exubera, it imposed limitations so the drug could not be prescribed for patients with asthma or other lung conditions, and Afrezza could face similar restrictions. An FDA staff report, issued just days before the advisory panel meeting, tied use of Afrezza to a decline in lung function and bronchial spasms that caused some patients to stop treatment. Restrictions on Afrezza could decrease the potential size of its market, which explains why not all analysts are exuberant. “With a high probability of an approval and a partnership sometime this summer, we believe expectations will be riding high. However, we expect Afrezza’s launch to be a slow one,” wrote Simos Simeonidis, an analyst at Cowen & Co. of New York, in a note April 2. “We continue to view commercialization as a significant challenge, especially in light of what we expect to be a label with a number of restrictions.” And if Afrezza reaches the commercial market, it is unclear how much market share it would steal from oral or injectable insulin. For type 2 diabetics, whose illness is often tied to poor dieting and lifestyle habits, Afrezza could offer a step in the process prior to a patient needing to move to undesirable injectables. “The most natural place where this could fit would be people on oral medications who don’t want to go onto injectable insulin,” Suvannavejh said. “Doctors will have to play with it to find out though. It will fit somewhere and they’ll figure it out over time.” But Pfeffer, MannKind’s CFO, thinks replacing injectable insulin is a long-term possibility. “There’s still a lot of work to do,” he said. “I think our product is potentially a replacement.” Economic recovery The financial success could not come too early for MannKind, which in the near-decade it has worked on Afrezza has reported consistent financial losses, staying alive almost entirely on the financial backing of Mann. In its fourth quarter earnings released in February, MannKind reported a loss of $53.6 million (-16 cents a share) for the fourth quarter ended Dec. 31, compared to a loss of $51.8 million (-23 cents) for the same quarter a year earlier. Analysts on average expected a loss of 14 cents. In total, the company has reported losses of more than $2 billion since its founding. The company is also developing a cancer vaccine. While the first step is gaining FDA approval and market share in the United States, Afezza could become a global product. Ahmed Enany, chief executive of the Southern California Biomedical Council in Los Angeles, believes the drug will have a large and growing market oversees, based on current health trends. “The market is huge and it will only get bigger, even internationally,” he said. “A large amount of people, especially in China and India, are becoming diabetic. The market is there, but the portion of it MannKind can capture is the question and it’s too soon to know. If this can replace sticking yourself with a needle a couple times a day, patients everywhere would prefer it.”